Back فوسفيت Arabic فوسفیت AZB Anión fosfito Spanish فسفیت Persian Fosfiitti Finnish Phosphite French Fosfito Italian 아인산염 Korean Fosfit Serbo-Croatian Phosphite SIMPLE

| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Phosphonate
| |||
| Systematic IUPAC name
Phosphite[1] | |||
| Identifiers | |||
3D model (JSmol)
|
| ||
| ChEBI | |||
| ChemSpider | |||
| 1618 | |||
| MeSH | Phosphorite | ||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
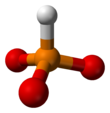
| HPO2− 3 | |||
| Molar mass | 79.9810 g mol−1 | ||
| Related compounds | |||
Other anions
|
Phosphinite | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
A phosphite anion or phosphite in inorganic chemistry usually refers to [HPO3]2− but includes [H2PO3]− ([HPO2(OH)]−). These anions are the conjugate bases of phosphorous acid (H3PO3). The corresponding salts, e.g. sodium phosphite (Na2HPO3) are reducing in character.
- ^ "Phosphorite - PubChem Public Chemical Database". The PubChem Project. USA: National Center for Biotechnology Information.